Research
- Identify epigenetic markers of early-life factors in association with childhood cancer risk (EpiEARLY)
- Identify epigenetic biomarkers of exposure in human populations and cancer risk and assess their validity in intervention studies (EpiMARKS)
- Identify and characterize epigenetic driver genes in cancer and their link to environmental carcinogens/exposures (EpiDRIVERS)
- Investigations of mutation spectra and their associated impact, and molecular markers of exposure, early disease, and early detection in experimental models and humans (MutSpec 2.0)
- Investigations of cancer causes through integrative molecular cancer epidemiology studies
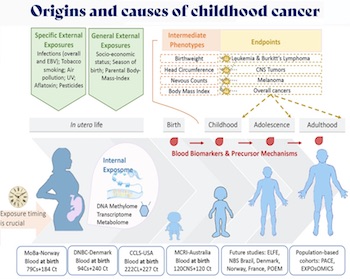
- The goal of this project is to understand the underlying molecular causes of cancers in children and adolescents by investigating epigenetic changes driven by risk factors and identifying epigenetic markers for cancer risk stratification and early detection. The hypothesis is that epigenome changes associated with risk of childhood cancer and exposure to environmental, dietary, and lifestyle factors during pregnancy can be identified in blood cells at birth, and that these methylation changes can serve as sensitive biomarkers in primary and secondary prevention as well as targeted therapy of childhood cancer.
- This project builds on EGM’s recent advances in identifying epigenetic markers of early-life factors and deciphering their precursor roles in childhood cancer and its predisposing (intermediate) phenotypes at critical ages of development, exploiting EGM’s large, unique, and complementary networks of prospective and retrospective consortia (Figure 5).
- The main goal of this project (EpiMARKS) is to test the hypothesis that specific epigenetic changes may be associated with and/or caused by specific dietary or lifestyle factors, as well as that these epigenetic patterns can serve as sensitive biomarkers of cancer outcomes and in monitoring the impact of interventions aimed at reducing cancer risk (primary and tertiary prevention) (Figure 6).
- The focus is on breast cancer and colorectal cancer, two common human cancer types that share major modifiable risk factors (such as lifestyle, obesity, alcohol consumption, and physical inactivity). Moreover, these cancer types have rising incidence rates in low- and middle-income countries and can be associated with particular risk factors that are increasingly present in those regions (e.g. changes in lifestyle and exposure to endocrine disrupters). The project encompasses two overarching aims in relation to breast cancer and colorectal cancer: (1) identifying epigenetic biomarkers of exposure and cancer risk (EpiSIGNATURES), and (2) examining the impact of interventions on epigenetic markers associated with cancer risk (EpiTRIALS). EGM is capitalizing on unique prospective cohorts (which enable the measurement of epigenetic events at different time points before diagnosis, typically in surrogate tissues such as blood), case–control studies (covering high-incidence regions and enabling the detection of epigenetic aberrations directly in the “tumour target” tissues), and intervention studies (which enable examination of the potential of targeted interventions for the purpose of reverting disease-associated epigenetic patterns as well as deciphering “causal” associations by direct experimentation).
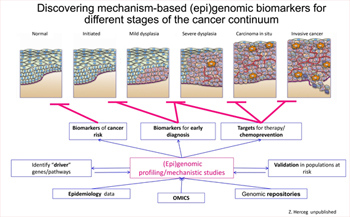
- Another focus in this area is aimed at identifying epigenetic markers of oesophageal squamous cell carcinoma, focusing on high-incidence populations of the world (EpiESCC). The goal of this project is to understand the underlying biological causes of oesophageal squamous cell carcinoma in high-incidence regions by investigating epigenetic (DNA methylation) aberrations driven by risk factors and identifying molecular markers for risk stratification and early detection. This study takes advantage of the unique epidemiological study, coordinated by IARC, of oesophageal squamous cell carcinoma in high-incidence populations, which provides a large series of tumour, tumour-adjacent normal, and surrogate tissue samples with high-quality epidemiological and clinical data.
- One of the most remarkable findings of the international high-resolution cancer genome sequencing efforts, spearheaded by the Cancer Genome Atlas, is the high frequency of genetic alterations in the genes encoding proteins that directly regulate the epigenome – referred to here as epigenetic regulator genes (ERGs) – in common human cancer types. This high rate of genetic deregulation of ERGs constitutes a “genetic smoking gun” that epigenetic mechanisms lie at the very heart of cancer biology. However, much remains unknown about the impact of these changes on the expression and function of ERGs.
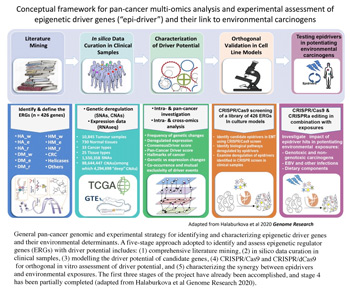
These findings have sparked debate on these genes potentially acting as “drivers” of tumorigenesis and on the mechanisms fuelling epigenome changes that are rampant in human malignancies. This study (EpiDRIVERS) aims to test the hypothesis that ERGs, when disrupted through genetic or non-mutational mechanisms, may act as drivers (“epidrivers”) in cancer development and confer a cancer cell phenotype (and potentially synergize with environmental exposure) (Figure 7). This project builds on EGM’s previous work that provided important conceptual advances on the role of epigenetic regulators in chromatin-based processes, including transcription and DNA repair.
- Recently, EGM developed and applied novel bioinformatics and statistical approaches, integrating the strengths of various driver prediction algorithms, which revealed ERGs with driver roles within and across malignancies. EGM further developed and tested a conceptual framework to identify and orthogonally validate in cellular models functionally important epidriver genes through a novel systematic approach that integrates the strengths of state-of-the-art genome-editing screens (CRISPR/Cas9). The results of the screen identified a panel of ERGs with a potential driver role conferring on cancer cells the traits associated with specific hallmarks of cancer. Further studies are focused on characterizing epidrivers that play an essential role in tumorigenesis and on investigating whether specific epidrivers have a central role in associating environmental exposures with cancer, including multiple phenotypic traits across the carcinogenesis process and using different biological systems. Answering this question should contribute to a better understanding of mechanisms of carcinogenesis and tumour classification, and provide important information for carcinogen evaluation and classification.
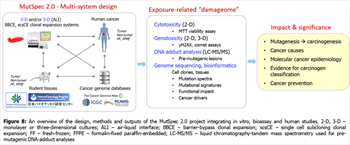
- Key research activities include experimental toxicogenomics studies of cancer risk agents (the MutSpec 2.0 project). These comprise the systematic genome-scale characterization of carcinogen exposure effects in experimental cell models, analyses of mutation spectra in tissues derived from animal bioassays and human tumour sample collections, and collaborative activities related to the prioritization of cancer risk agents for mutagenicity testing (Figure 8).
- Innovative experimental systems and methodologies are designed and applied within the MutSpec 2.0 project. The current focus is on the advancement of established model systems to develop rapid and high-throughput applications for carcinogen testing and to study mechanisms of DNA damage, including collaborative adductomic approaches to establish the formation of pre-mutagenic lesions. The functional impact of mutagenic insults is investigated by identifying the relationships between mutagenesis and genome topography, by analysing mutation spectra and the dynamics of chromatin organization during cell immortalization and transformation and in cancer. A particular focus is on the functional interplay between genetic and epigenetic alterations during early stages of cell transformation, as well as the validation of the obtained experimental molecular markers in the public pan-cancer data.
- By providing expertise in genomics and bioinformatics related to genotoxic insults by suspected or known carcinogens, EGM participates in various molecular cancer epidemiology studies, including (but not limited to) investigations of renal carcinogenicity linked to dietary exposure to acrylamide (the MODARC project, supported by World Cancer Research Fund International), the role of asbestos exposure in the development of ovarian cancer (the EVAMOVAIRE2 study, supported by ANSES, France), metabolic and genetic signatures in triple-negative breast cancer from the EPIC cohort (supported by INCa, France), and exposure to the herbal carcinogen aristolochic acid and the associated cancer risk in Asia and Europe. In the settings of low- and middle-income countries, EGM collaborates on investigations of the molecular underpinnings of regional high-incidence cancer types (ARISTOCANCERS project). The results from model-based mechanistic and molecular epidemiology studies are translated into biomarkers of exposure and cancer development that are potentially applicable to cancer hazard assessment.